Introduction
Heat transfer is an important chapter in ICSE Class 8 Physics. It explains how heat moves from one body to another and helps students understand daily life examples like cooking, boiling water, sea breeze, room heaters, and refrigerators. If you are searching for ICSE Class 8 Physics Heat Transfer Notes PDF Download, this article provides clear notes, definitions, examples, and formulas in simple language.
These notes are prepared according to the latest ICSE syllabus and are useful for quick revision, homework, and exam preparation.
Study Support by Rohit Academy
Rohit Academy offers expert-curated ICSE Class 8 Physics Study Materials including ICSE Heat Transfer Chapter Notes, diagrams, and key formulas for better understanding.
You can also visit:
| ☛ ICSE Class 8 Physics Chapter 6: Heat Transfer Selina Solutions |
| ☛ ICSE Class 8 Physics |
| ☛ ICSE Class 8 Chemistry |
| ☛ ICSE Class 8 Mathematics |
| ☛ ICSE Class 8 Biology |
Heat Transfer Notes ICSE Class 8
Introduction
- Matter is anything that has mass and occupies space.
- Matter is made up of tiny particles called molecules.
- Molecules:
- Are extremely small.
- Are in continuous motion.
- Have kinetic energy due to motion.
- Have potential energy due to forces of attraction.
Energy in Different States
- Solid → Highest potential energy, least kinetic energy
- Gas → Least potential energy, greatest kinetic energy
Heat and Molecular Motion
- Heat is a form of energy that flows from a hot body to a cold body when they are kept in contact.
- Unit of Heat: Joule (J)
- When a substance is heated:
- Molecules move faster.
- Kinetic energy increases.
- When a substance is cooled:
- Molecular motion slows down.
- Kinetic energy decreases.
Internal Energy
- The total kinetic energy of molecules = Internal kinetic energy.
- The total potential energy of molecules = Internal potential energy.
- The sum of both is called internal energy (heat energy).
Flow of Heat
- Heat always flows from:
- Higher temperature → Lower temperature
- Temperature depends on the average kinetic energy of molecules.
EFFECTS OF HEAT
Heat mainly produces:
- Change in temperature of a body
- Change in state of matter
- Change in size of a body (expansion)
1. Change in Temperature
- When a body is heated:
- Temperature rises.
- When a body is cooled:
- Temperature falls.
Factors Affecting Temperature Rise
- Quantity of heat supplied
- Nature of material (specific heat capacity)
2. Change in State of Matter
The process of changing from one state of matter to another at constant temperature is called change of state.
Matter exists in three states:
- Solid
- Liquid
- Gas
Examples of Change of State

- Solid → Liquid (Melting)
- Liquid → Gas (Boiling / Vaporization)
- Gas → Liquid (Condensation)
- Liquid → Solid (Freezing / Solidification)
- Solid → Gas directly (Sublimation)
Melting (Fusion)
- Change of solid into liquid at fixed temperature is called melting or fusion.
- The temperature at which melting occurs is called melting point.
- Example: Melting point of ice is 0°C.
Vaporization (Boiling)
- Change of liquid into vapour at fixed temperature is called vaporization or boiling.
- Temperature remains constant during boiling.
- Example: Water boils at 100°C
Freezing (Solidification)
- Change of liquid into solid on cooling is called solidification or freezing.
- Example: Freezing point of water is 0°C.
Condensation
- Vapour changes into liquid on cooling.
Sublimation
- Some solids change directly into vapour on heating.
- Vapour changes back into solid on cooling.
- Examples: Camphor, Naphthalene
Latent Heat
- Heat absorbed or released during change of state without change in temperature is called latent heat.
- It is used to overcome forces of attraction between molecules.
Important Points
- Temperature remains constant.
- Heat is used to overcome forces of attraction between molecules.
- Kinetic energy remains constant.
- Potential energy changes.
EVAPORATION
- Evaporation is the change of liquid into vapour at any temperature from the surface is called evaporation.
- Example:
- A drop of ether or perfume placed on the palm disappears quickly.
- The palm feels cool because evaporation absorbs heat.
Nature of Evaporation
- It is a surface phenomenon.
- Occurs at all temperatures.
- It can be explained using molecular motion.
Explanation of Evaporation (Molecular Level):
- Molecules in a liquid are in motion.
- Some molecules at the surface gain higher kinetic energy.
- only the molecules at the surface overcome the forces of attraction and escape into air.
- The escaping molecules form vapour.
- The process continues until the liquid evaporates.
- Energy required is taken from surroundings → cooling effect.
Factors Affecting Rate of Evaporation
1. Temperature
- Higher temperature → faster evaporation.
- Reason: Molecules gain more kinetic energy.
2. Blowing Air (Wind)
- Wind increases evaporation.
- Reason: Moving air removes vapour molecules from the surface, allowing more molecules to escape.
3. Surface Area
- Larger surface area → faster evaporation.
- Reason: More molecules are exposed to air.
4. Humidity
- Higher humidity → slower evaporation.
- Reason: Air already contains water vapour, so fewer molecules escape.
Cooling Produced During Evaporation
- Explanation
- As the liquid absorbs heat, the surroundings lose heat.
- Loss of heat makes the surroundings cooler.
- Hence, evaporation produces a cooling effect.
- Example
- Spirit or ether on the skin produces a cooling effect.
- Water in an earthen pot remains cool due to evaporation.
- Sweating cools our body when sweat evaporates.
BOILING
- Boiling is the change of liquid into vapour at a fixed temperature.
Boiling Point
- The boiling point is the temperature at which a liquid changes into vapour.
- Example:
- Water boils at 100°C at normal atmospheric pressure.
Characteristics of Boiling
- Occurs throughout the liquid.
- Bubbles form inside the liquid.
- Temperature remains constant during boiling.
Explanation (Molecular Level)
- Molecules gain energy and escape throughout the liquid.
- During boiling, vaporization occurs throughout the liquid.
- Bubbles form and rise to surface.
Average Kinetic Energy at Boiling Point
- Temperature is a measure of the average kinetic energy of molecules.
- During boiling, the temperature remains constant.
- Since temperature does not increase, the average kinetic energy of molecules does not change.
Effect of Pressure on Boiling Point
- Increase in pressure → boiling point increases
- Decrease in pressure → boiling point decreases
- Example: Water boils at lower temperature in mountains.
Difference Between Evaporation and Boiling
| Evaporation | Boiling |
| Evaporation occurs at all temperatures. | Boiling occurs at a fixed temperature. |
| Evaporation is a slow process. | Boiling is a rapid process. |
| Evaporation is a surface phenomenon. | Boiling occurs throughout the liquid. |
| Causes cooling | Does not produce cooling in the same way |
| No bubble formation | Bubbles form |
Absolute Zero
- As temperature decreases, molecular motion slows down.
- At a certain extremely low temperature, molecular motion completely stops.
- This temperature is called absolute zero.
- Absolute zero on the Kelvin scale = 0 K.
- Temperatures lower than absolute zero are not possible.
THERMAL EXPANSION
Thermal expansion is the increase in size of a substance when heated and contraction when cooled.
Reason (Molecular Explanation)
- On heating, molecules gain kinetic energy.
- They vibrate faster and move farther apart.
- Intermolecular distance increases.
- As a result, the substance expands.
Expansion in Different States
Solids
- Particles are tightly packed.
- Expansion is small.
- Example: Railway tracks have small gaps to prevent bending in summer.
Liquids
- Particles are less tightly packed.
- Liquids expand more than solids.
- Example: Liquid in thermometers rises when heated.
Gases
- Particles are far apart.
- Gases expand much more than solids and liquids.
Thermal Expansion in Solids
- A solid has a definite shape.
- When heated:
- Length increases
- Area increases
- Volume increases
- Types of expansion in solids:
- Linear expansion – Increase in length
- Superficial expansion – Increase in area
- Cubical expansion – Increase in volume
Demonstration of Thermal Expansion (Ball and Ring Experiment)
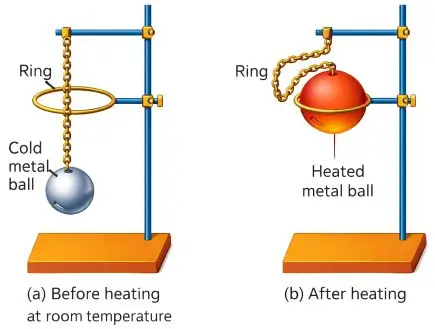
Procedure
- A metal ball fits through a ring at room temperature.
- Heat the ball.
- Try passing it through the ring again.
Observation
- The heated ball does not pass through the ring.
Conclusion
- The ball expands on heating.
- After cooling, the ball contracts and passes through again.
Bar and Gauge Experiment

Observation
- A metal bar fits in a gauge at room temperature.
- On heating, it does not fit.
- After cooling, it fits again.
Conclusion
- Solids expand on heating and contract on cooling.
Linear Expansion
When a solid in the form of a rod or wire is heated, only the increase in length is noticeable.
This is called linear expansion.
Factors Affecting Linear Expansion
The increase in length of a rod depends on:
- Original length of the rod
- Longer rods expand more.
- Rise in temperature
- Higher temperature → more expansion.
- Material of the rod
- Different materials expand by different amounts.
- Example: Copper expands more than iron.
Formula of Linear Expansion
If
- \(L_0=\) original length
- \(L_t=\) length at temperature, t
- \(\alpha\ =\) coefficient of linear expansion
Then: \(\operatorname{L}_t\ -\operatorname{L}_0=\operatorname{L}_0\ \alpha\ t\)
Coefficient of Linear Expansion
- Depends on the material of the rod.
- Unit: per °C
Important Fact
- The expansion of a rod does not depend on whether it is hollow or solid.
- Rods of the same material and length expand equally for the same temperature rise.
Bimetallic Strip (Application)
A bimetallic strip:
- Is made of two different metals joined together.
- When heated, one metal expands more than the other.
- This causes bending.
Example:
Brass expands more than iron, so the strip bends toward the iron side.
Superficial Expansion of Solids
When a metal plate is heated:
- Length increases
- Breadth increases
- Area increases
This increase in area is called superficial expansion.
Factors Affecting Superficial Expansion
- Initial area of plate
- Rise in temperature
- Material of the plate
Formula
If
- \(A_0=\) initial area
- \(A_t=\) area at temperature, t
- \(\beta=\) coefficient of superficial expansion
Then: \(\operatorname{A}_t-\operatorname{A}_0=\operatorname{A}_0\ \beta\ t\)
Cubical Expansion of Solids
When a solid is heated:
- Length increases
- Breadth increases
- Thickness increases
- Volume increases
This increase in volume is called cubical expansion.
Factors Affecting Cubical Expansion
- Initial volume
- Rise in temperature
- Material of the solid
Formula
If
- \(V_0=\) initial volume
- \(V_t=\) volume at temperature, t
- \(\delta=\) coefficient of cubical expansion
Then: \(V_t\ -V_0=V_0\ \gamma\ t\)
Relationship Between Expansion Coefficients
Relation:
β = 2α
γ = 3 α
Ratio:
α : β : γ = 1 : 2 : 3
Where:
- α = linear expansion
- β = superficial expansion
- γ = cubical expansion
APPLICATIONS OF THERMAL EXPANSION (SOLIDS)
1. Bridges
Rollers are used to allow expansion.
2. Railway Tracks
Gaps are left between rails to prevent bending in summer.
Reason: Metals expand on heating.
3. Riveting
Hot rivets contract on cooling and hold metal plates tightly.
Used in bridges, boilers, girders.
4. Electric Wires
Loose in summer, tight in winter.
Prevents breaking due to expansion and contraction.
5. Steel Rim of Wheel
Heated rim expands, fitted, then tightens on cooling.
6. Glassware (Pyrex)
Low expansion prevents cracking.
7. Pendulum (Invar)
Very small expansion maintains accuracy.
8. Loosening Metal Caps
Heating expands metal cap making it easier to open.
9. Cracking of Thick Glass
Unequal expansion of inner and outer surfaces causes cracks.
THERMAL EXPANSION IN LIQUIDS
- Liquids expand more than solids.
- Liquids have only cubical expansion.
Anomalous Expansion of Water
Water behaves differently from most liquids.
Behaviour of Water
- From 0°C to 4°C → Water contracts on heating.
- Above 4°C → Water expands normally.
This is called anomalous expansion of water.
Important Fact
Water has maximum density at 4°C.
Explanation of Expansion of Liquids (Molecular View)
- In a liquid, molecules are free to move.
- On heating:
- Average kinetic energy of molecules increases.
- Molecules move more vigorously.
- Intermolecular distance increases.
- As a result, the liquid expands.
Factors Affecting Expansion of Liquids
The cubical expansion of a liquid depends on:
- Original Volume of Liquid
- Larger initial volume → greater expansion.
- Rise in Temperature
- Higher temperature rise → greater expansion.
- Nature of Liquid
- Different liquids expand by different amounts.
Experiment to Show Expansion of Liquid
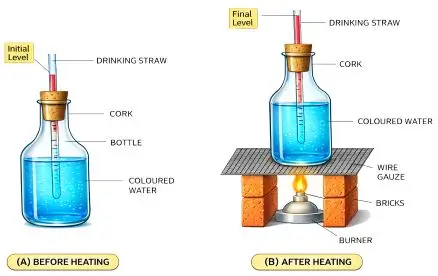
Procedure
- Take a bottle fitted with a cork and straw.
- Fill it with coloured water.
- Heat the bottle.
Observation
- Water level in the straw rises.
Conclusion
- Liquids expand on heating.
Formula for Cubical Expansion of Liquids
If
- \(V_0=\) initial volume
- \(V_t=\) volume at temperature, t
- \( \delta=\) coefficient of cubical expansion
Then: \(V_t\ -V_0=V_0\ \gamma\ t\)
APPLICATIONS OF EXPANSION OF LIQUIDS
- Mercury thermometer works on expansion of mercury.
Different Liquids Expand Differently
Equal volumes of different liquids heated to the same temperature expand by different amounts.
Example Order of Expansion
- Benzene expands most
- Alcohol and kerosene expand moderately
- Water expands least
THERMAL EXPANSION IN GASES
- Gases expand more than solids and liquids.
- Heating increases molecular motion and separation.
Demonstration of Expansion of Gases
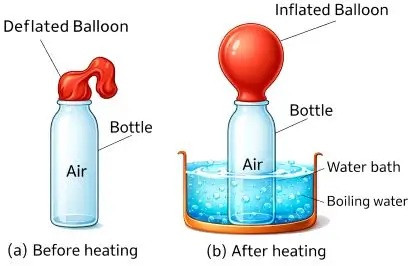
Activity
- Take an empty bottle and attach a balloon.
- Heat the bottle in warm water.
Observation
- Balloon inflates.
Conclusion
- Air expands on heating.
VARIATION OF DENSITY WITH TEMPERATURE
- On heating:
- Mass remains constant
- Volume increases
- Density decreases
- Exception:
- Water density increases from 0°C to 4°C.
EFFECT OF TEMPERATURE ON MOLECULAR MOTION
- Higher temperature → faster molecular motion
- Lower temperature → slower molecular motion
- At absolute zero, molecular motion theoretically stops.
ICSE Class 8 Physics Heat Transfer Notes PDF Download
Students can use these notes for revision before exams. These notes are written in easy language for quick understanding and better marks.
Conclusion
This chapter helps students understand how heat moves in solids, liquids, gases, and even through empty space. Practice definitions and examples regularly to score well in exams.
ICSE Class 8 Physics Notes
Official Website:
Students can visit the official CISCE website for more details and updates.



