Introduction
Physics becomes more interesting when we understand how heat moves from one object to another. ICSE Class 8 Physics Chapter 6 Heat Transfer Selina Solutions helps students learn important concepts like conduction, convection, radiation, conductors, insulators, and sea breeze in a simple way. These step-by-step solutions are designed according to the latest ICSE syllabus and Selina textbook pattern.
Students often find numerical problems and theory questions from this chapter challenging. With these Selina Solutions for ICSE Class 8 Physics Chapter 6 Heat Transfer, you can easily understand each answer with proper explanation. It also helps improve problem-solving skills and prepares students for school exams.
Why Students Need These Solutions
Many students search for trusted answers before exams. These ICSE Class 8 Physics Chapter 6 Heat Transfer Selina Solutions provide clear explanations and correct answers to all exercise questions. Students can revise quickly and score better marks.
Study Support by Rohit Academy
Rohit Academy offers expert-curated ICSE Class 8 Physics Study Materials including ICSE Heat Transfer Chapter Notes, diagrams, and key formulas for better understanding.
You can also visit:
| ☛ ICSE Class 8 Physics Chapter 6: Heat Transfer Notes |
| ☛ ICSE Class 8 Physics |
| ☛ ICSE Class 8 Chemistry |
| ☛ ICSE Class 8 Mathematics |
| ☛ ICSE Class 8 Biology |
(A) Multiple Choice Type
Question 1
Choose the correct answer from the multiple choices given below.
Question 1(a)
In evaporation:
(i) all molecules of liquid begin to escape out
(ii) only the molecules at the surface escape out
(iii) the temperature of liquid rises by absorbing heat from surroundings
(iv) the molecules get attracted within the liquid
Answer:
(ii) only the molecules at the surface escape out
Explanation:
Evaporation is a surface phenomenon. Only the molecules at the surface that have enough kinetic energy escape into vapour.
Question 1(b)
The rate of evaporation of a liquid increases when:
(i) temperature of liquid falls
(ii) liquid is poured in a vessel of less surface area
(iii) air is blown above the surface of liquid
(iv) humidity increases
Answer:
(iii) air is blown above the surface of liquid
Explanation:
Blowing air removes vapour near the surface, increasing the rate of evaporation. Lower temperature, less surface area, and higher humidity all decrease evaporation.
Question 1(c)
During boiling or vaporization:
(i) all molecules take part
(ii) temperature rises
(iii) no heat is absorbed
(iv) the average kinetic energy of molecules increases
Answer:
(i) all molecules take part
Explanation:
During boiling, vaporization occurs throughout the liquid, not just at the surface. The temperature remains constant and heat is absorbed as latent heat, so the average kinetic energy does not increase.
Question 1(d)
The boiling point of a liquid is increased by:
(i) increasing the volume of liquid
(ii) increasing the pressure on liquid
(iii) adding ice to the liquid
(iv) decreasing pressure on liquid
Answer:
(ii) increasing the pressure on liquid
Explanation: A higher external pressure makes it harder for bubbles to form, so the boiling point increases.
Question 1(e)
Two rods A and B of the same metal, but of length 1 m and 2 m respectively, are heated from 0°C to 100°C. Then:
(i) both the rods A and B elongate the same
(ii) the rod A elongates more than the rod B
(iii) the rod B elongates more than the rod A
(iv) the rod A elongates, but the rod B contracts
Answer:
(iii) the rod B elongates more than the rod A
Explanation:
Expansion of a rod is proportional to its original length. Since rod B is longer, it expands more.
Question 1(f)
Two rods A and B of the same metal, same length, but one solid and other hollow, are heated to the same rise in temperature Then:
(i) the solid rod A expands more than the hollow rod B
(ii) the hollow rod B expands more than the solid rod A
(iii) the hollow rod B contracts, but the solid rod A expands
(iv) both the rods A and B expand the same
Answer:
(iv) both the rods A and B expand the same
Explanation:
Linear expansion depends on material, original length, and temperature rise—not on whether the rod is solid or hollow.
Question 1(g)
A given volume of alcohol and the same volume of water are heated from the room temperature to the same temperature then:
(i) alcohol contracts, but water expands
(ii) water contracts, but alcohol expands
(iii) water expands more than alcohol
(iv) alcohol expands more than water
Answer:
(iv) alcohol expands more than water
Explanation:
Alcohol has a higher coefficient of expansion than water, so for the same temperature rise it expands more.
Question 1(h)
The increase in length of a metal rod depends on:
(i) the initial length of the rod only
(ii) the rise in temperature only
(iii) the material of the rod only
(iv) all the above three factors
Answer:
(iv) all the above three factors
Explanation:
Increase in length depends on initial length, rise in temperature, and the material (coefficient of expansion).
Question 1(i)
The correct statement is:
(i) Iron rims are cooled before they are placed on the cart wheels.
(ii) A glass stopper gets tightened on warming the neck of the bottle.
(iii) Telephone wires sag in winter, but become tight in summer.
(iv) A little space is left between two rails on a railway track.
Answer:
(iv) A little space is left between two rails on a railway track.
Explanation:
Metals expand on heating; gaps are left so rails can expand in hot weather without bending.
Assertion Reason Type
Question 2
The following questions are Assertion–Reason based questions. Choose the answers based on the code given below.
Question 2(a)
Assertion (A) : A humid environment slows the rate of evaporation.
Reason (R) : In humid environment, vapour molecules do not find the space to escape.
(i) Both A and R are true and R is the correct explanation of A
(ii) Both A and R are true and R is not the correct explanation of A
(iii) Assertion is true but reason is false
(iv) Assertion is false but reason is true
Answer:
(i) Both A and R are true and R is the correct explanation of A
Explanation:
In a humid environment, the air already contains a large amount of water vapour. Since fewer vapour molecules can escape into the air, the rate of evaporation slows down.
Question 2(b)
Assertion (A) : Glass containers with narrow necks are more likely to break when hot water is poured into them suddenly.
Reason (R) : Glass is a good conductor of heat.
(i) Both A and R are true and R is the correct explanation of A
(ii) Both A and R are true and R is not the correct explanation of A
(iii) Assertion is true but reason is false
(iv) Assertion is false but reason is true
Answer:
(iii) Assertion is true but reason is false
Explanation:
The assertion is true because when hot water is poured suddenly, the inner surface of the glass expands quickly while the outer surface remains cooler, causing unequal expansion and possible cracking. The reason is false because glass is a poor conductor of heat, not a good conductor.
Question 2(c)
Assertion (A) : A gas expands less than a liquid for the same rise in temperature.
Reason (R) : Gases have negligible intermolecular forces of attraction.
(i) Both A and R are true and R is the correct explanation of A
(ii) Both A and R are true and R is not the correct explanation of A
(iii) Assertion is true but reason is false
(iv) Assertion is false but reason is true
Answer:
(iv) Assertion is false but reason is true
Explanation:
The assertion is false because gases expand more than liquids for the same rise in temperature. The reason is true: gases have negligible intermolecular forces, allowing them to expand easily.
Fill in the blanks
Question 3
Fill in the blanks:
(a) Boiling occurs at …………… .
(b) Evaporation takes place at …………… .
(c) The molecules of liquid …………… heat from surroundings in evaporation.
(d) Heat is …………… during boiling.
(e) Cooling is produced in …………… .
(f) A longer rod expands …………… than a shorter rod on being heated to the same temperature.
(g) Liquids expand …………… than the solids.
(h) Gases expand …………… than the liquids.
(i) Alcohol expands …………… than water.
(j) Iron expands …………… than copper.
Answer:
(a) Boiling occurs at a fixed temperature.
(b) Evaporation takes place at all temperatures.
(c) The molecules of liquid absorb heat from surroundings in evaporation.
(d) Heat is absorbed during boiling.
(e) Cooling is produced in evaporation.
(f) A longer rod expands more than a shorter rod on being heated to the same temperature.
(g) Liquids expand more than the solids.
(h) Gases expand more than the liquids.
(i) Alcohol expands more than water.
(j) Iron expands less than copper.
True or False
Question 4
Write true or false for each statement:
(a) Evaporation is rapid on a wet day.
(b) Evaporation takes place only from the surface of a liquid.
(c) All molecules of a liquid take part in the process of evaporation.
(d) Temperature of a liquid rises during boiling or vaporization.
(e) All molecules of a liquid take part in boiling.
(f) Boiling is a rapid phenomenon.
(g) All solids expand by the same amount when heated to the same rise in temperature.
(h) Telephone wires are kept tight between the two poles in winter.
(i) Equal volumes of different liquids expand by different amounts when they are heated to the same rise in temperature.
(j) Solids expand the least and gases expand the most on being heated.
(k) A mercury thermometer makes use of the property of expansion of liquids on heating.
(l) Kerosene contracts on heating.
Answer:
(a) False
Correct Statement : Evaporation is rapid on a dry day.
(b) True
(c) False
Correct Statement : Only the molecules on the surface of a liquid take part in the process of evaporation.
(d) False
Correct Statement : Temperature of a liquid remains constant during boiling or vaporization.
(e) True
(f) True
(g) False
Correct Statement : All solids do not expand by the same amount when heated to the same rise in temperature.
(h) True
(i) True
(j) True
(k) True
(l) False
Correct Statement : Kerosene expands on heating.
Match the Columns
Question 5
Match the following:
| Column A | Column B |
| (a) Blowing air increases | (i) increase in inter-molecular separation |
| (b) Increase in pressure increases | (ii) pendulum of a clock |
| (c) Thermal expansion | (iii) cooking utensils |
| (d) Invar | (iv) boiling point |
| (e) Pyrex glass | (v) evaporation |
Answer:
| Column A | Column B |
| (a) Blowing air increases | (v) evaporation |
| (b) Increase in pressure increases | (iv) boiling point |
| (c) Thermal expansion | (i) increase in inter-molecular separation |
| (d) Invar | (ii) pendulum of a clock |
| (e) Pyrex glass | (iii) cooking utensils |
(B) Short Answer Type Questions
Question 1
What is matter? What is it composed of?
Answer:
Matter is anything that has mass and occupies space. Matter is composed of very tiny particles called molecules (or atoms).
Question 2
Do all the molecules of a liquid take part in evaporation? If not, explain your answer.
Answer:
No, all molecules do not take part in evaporation. Only the molecules at the surface of the liquid that have sufficient kinetic energy escape into vapour. Molecules inside the liquid cannot escape because they are surrounded by other molecules.
Question 3
No heat is supplied to a liquid during evaporation. How does then the liquid change into its vapour?
Answer:
During evaporation, the liquid absorbs heat from the surroundings. This heat provides the energy needed for surface molecules to escape into vapour.
Question 4
Comment on the statement ‘Evaporation is a surface phenomenon’.
Answer:
Evaporation occurs only at the surface of a liquid because only surface molecules can overcome intermolecular forces and escape into vapour. Molecules inside the liquid cannot evaporate directly.
Question 5
Why is cooling produced when a liquid evaporates.
Answer:
During evaporation, a liquid changes into vapour and requires heat for this process. Since no external heat is supplied, the liquid absorbs the required heat from its surroundings, which produces a cooling effect.
Question 6
What is the change in average kinetic energy of molecules of a liquid during boiling at its boiling point?
Answer:
There is no change in the average kinetic energy of the molecules because the temperature remains constant at the boiling point.
Question 7
How is the heat energy supplied to a liquid used during boiling at a fixed temperature?
Answer:
The heat supplied during boiling is used to overcome intermolecular forces and convert the liquid into vapour. This heat is called latent heat of vaporization.
Question 8
Name two ways of changing liquid state to the vapour state and distinguish them.
Answer:
The two ways to change liquid state to vapour state are:
- Boiling
- Evaporation
Differences between Boiling and Evaporation:
| Boiling | Evaporation |
| Boiling occurs at a fixed temperature. | Evaporation occurs at all temperatures. |
| Boiling is a rapid process. | Evaporation is a slow process. |
| Boiling occurs throughout the liquid. | Evaporation is a surface phenomenon. |
| In boiling, external heat is supplied. | In evaporation, liquid molecules absorb heat from its surroundings to convert into vapour state. |
Question 9
What do you understand by thermal expansion of a substance?
Answer:
Thermal expansion is the increase in size (length, area, or volume) of a substance when its temperature is increased.
Question 10
Give two examples of the substances which expand on heating.
Answer:
Iron and alcohol expand on heating.
Question 11
State three factors on which depend the linear expansion of a metal rod on heating.
Answer:
Linear expansion depends on:
- Original length of the rod
- Rise in temperature
- Nature of material
Question 12
A metal plate is heated. State three factors on which the increase in its area will depend.
Answer:
Increase in area depends on:
- Original area of the plate
- Rise in temperature
- Nature of material
Question 13 A cubical metal solid block is heated. How will its volume change?
Answer:
When a cubical metal block is heated, its length, breadth, and thickness increase, and therefore its volume also increases.
Question 14
State one application of thermal expansion of liquids.
Answer:
Thermal expansion of liquids is used in the working of mercury thermometer.
Question 15
Which of the following will expand more, when heated to the same temperature:
(a) solid, (b) liquid or (c) gas?
Answer:
Gas expands more than solids and liquids because the intermolecular forces of attraction between gas molecules are very weak, and the molecules have high kinetic energy, allowing them to move farther apart on heating.
Question 16
100 ml of each of the following liquids is heated from 10°C to 50°C. Which will expand more:
(a) water (b) benzene (c) alcohol?
Answer:
Benzene will expand the most.
Question 17
Water is heated from 0°C to 4°C. Will it expand?
Answer:
No, Water contracts when heated from 0°C to 4°C.
Question 18
What do you mean by anomalous behavior of water?
Answer:
The anomalous behavior of water refers to the fact that water contracts when heated from 0°C to 4°C and expands when cooled from 4°C to 0°C. Water has maximum density at 4°C.
Question 19
How does the density of a substance (solid, liquid and gas) change on heating?
Answer:
On heating, the density of solids, liquids, and gases decreases.
(C) Long Answer Type Questions
Question 1
Name the three states of matter and distinguish them on the basis of their (i) volume, and (ii) shape
Answer:
Three states of matter are solid, liquid, and gas.
| State | Volume | Shape |
| Solid | Definite | Definite |
| Liquid | Definite | No definite shape |
| Gas | No definite volume | No definite shape |
Question 2
Distinguish between liquid and vapour (or gas) states of matter on the basis of the following factors:
(a) Arrangement of molecules
(b) Inter-molecular separation
(c) Inter-molecular force and
(d) Kinetic energy of molecules
Answer:
| Factor | Liquid | Vapour / Gas |
| Arrangement of molecules | Molecules are loosely packed | Molecules are very far apart |
| Intermolecular separation | Small | Very large |
| Intermolecular force | Moderate | Very weak or negligible |
| Kinetic energy of molecules | Less than gases | Very high |
Question 3
What is evaporation? Explain it on the basis of molecular motion.
Answer:
Evaporation is the change of a liquid into vapour at any temperature below its boiling point.
Explanation:
Molecules in a liquid are in constant motion. Some molecules at the surface gain sufficient kinetic energy to overcome intermolecular forces and escape into vapour. Hence evaporation occurs only at the surface.
Question 4
What is boiling? Explain it on the basis of molecular motion.
Answer:
Boiling is the change of a liquid into vapour on heating at a constant temperature.
When a liquid is heated, the average kinetic energy of its molecules increases. Once the molecules gain enough energy to overcome the intermolecular forces of attraction, they escape as vapour throughout the liquid, not just from the surface.
Question 5
Describe an experiment to demonstrate the thermal expansion in solids.
Answer:
Experiment:
- Take a metal ball and ring apparatus.
- At room temperature, the ball passes easily through the ring.
- Heat the metal ball and try to pass it through the ring again.
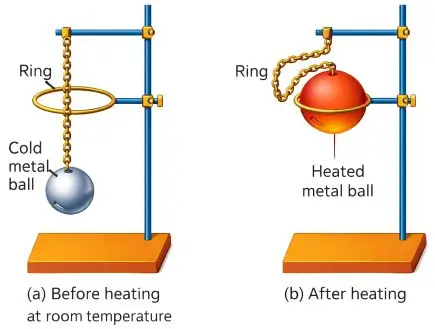
Observation:
The heated ball does not pass through the ring.
Conclusion:
The metal ball expands on heating, showing thermal expansion in solids.
Question 6
Describe an experiment to show that liquids expand on heating.
Answer:
Experiment:
- Take a flask filled with coloured water and fit a glass tube through a cork.
- Mark the initial level of water in the tube.
- Heat the flask gently.
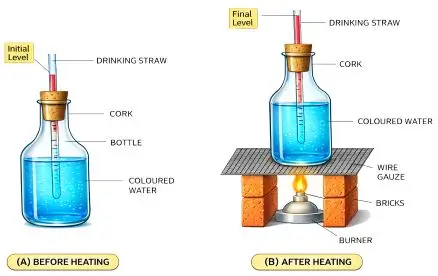
Observation:
The level of liquid in the tube rises.
Conclusion:
Liquids expand on heating.
Question 7
Describe an experiment to show that air expands on heating.
Answer:
Experiment:
- Take a dry bottle and fix a balloon over its mouth.
- Place the bottle in a container of hot water.
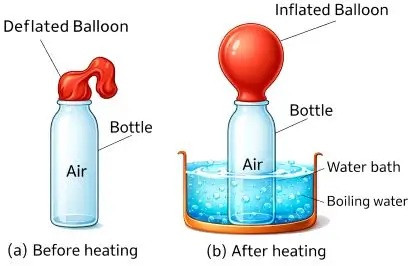
Observation:
The balloon inflates.
Conclusion:
Air inside the bottle expands on heating and fills the balloon.
Question 8
Describe an experiment to show that same volume of different liquids heated to same rise in temperature expand by different amounts.
Answer:
Experiment:
Take four identical glass bottles fitted with narrow glass tubes through corks. Fill each bottle to the same level with different liquids such as water, alcohol, kerosene, and benzene. Place all the bottles in a common water bath containing boiling water and heat them for some time.
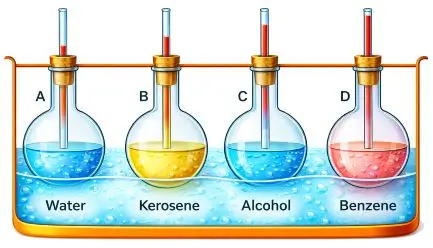
Observation:
After some time, the liquids rise in the tubes to different levels. Benzene rises the most, followed by alcohol and kerosene, while water rises the least.
Conclusion:
This experiment shows that equal volumes of different liquids, when heated through the same temperature range, expand by different amounts.
(D) Think and Answer
Question 1
Give reason for the increase in rate of evaporation of a liquid when:
(a) air is blown above the liquid.
(b) surface area of liquid is increased.
(c) temperature of liquid is increased.
Answer:
(a) Blowing air removes the vapour formed near the surface of the liquid, so more molecules can escape, increasing the rate of evaporation.
(b) A larger surface area exposes more molecules at the surface, allowing more molecules to escape at the same time, so evaporation increases.
(c) When temperature increases, the kinetic energy of molecules increases. More molecules gain enough energy to escape from the surface, so the rate of evaporation increases.
Question 2
Why does bubbles appear when a liquid is heated?
Answer:
When a liquid is heated, the average kinetic energy of its molecules increases. When the molecules gain enough energy to overcome the intermolecular forces of attraction, they escape as vapour throughout the liquid. As a result, bubbles are formed, which rise to the surface, and boiling takes place.
Question 3
Two iron rods — one 10 m long and the other 5 m long, are heated to the same rise in temperature. Which will expand more?
Answer:
The 10 m long iron rod will expand more.
Reason:
The increase in length of a rod is directly proportional to its original length. Since the 10 m rod is longer than the 5 m rod, it undergoes greater expansion for the same rise in temperature.
Question 4
Two identical rods of copper are heated to different temperatures — one by 5°C and the other by 10°C. Which rod will expand more?
Answer:
The rod heated by 10°C will expand more.
Reason:
The increase in length of a rod is directly proportional to the rise in temperature. Since the second rod experiences a greater temperature rise, it undergoes greater expansion.
Question 5
One rod of copper and another identical rod of iron are heated to the same rise in temperature. Which rod will expand more? Give reason.
Answer:
The copper rod will expand more.
Reason:
For the same initial length and rise in temperature, expansion depends on the coefficient of linear expansion of the material. Copper has a higher coefficient of linear expansion than iron, so it expands more when heated.
Question 6
Two identical rods — one hollow and the other solid, are heated to the same rise in temperature. Which will expand more?
Answer:
Both the rods will expand by the same amount, because the increase in length on heating does not depend on whether the rod is hollow or solid.
Question 7
In the ball and ring experiment, if the ball after heating is left to cool on the ring for some time, the ball again passes through the ring. Explain the reason.
Answer:
When the heated ball is left on the ring for some time, it loses heat to the surroundings and cools down. On cooling, the ball contracts and returns nearly to its original size. Therefore, it again passes through the ring.
Question 8
Explain the following:
(a) The telephone wires break in winter.
(b) Iron rims are heated before they are fixed on the wooden wheels.
(c) Gaps are left between the successive rails on a railway track.
(d) A glass stopper stuck in the neck of a bottle can be removed by pouring hot water on the neck of the bottle.
(e) A cement floor is laid in small pieces with gaps in between.
Answer:
(a) In winter, the temperature falls andmetal wires contract. This contraction increases the tension in the wires, which may cause them to snap or break.
(b) When heated, the iron rim expands and can be easily fitted over the wooden wheel. On cooling, it contracts and fits tightly, holding the wheel firmly.
(c) Rails expand in hot weather. Gaps are left to provide space for expansion, preventing the rails from bending or buckling.
(d) Hot water heats the neck of the bottle, causing it to expand slightly. The stopper expands less, so it becomes loose and can be removed.
(e) Cement expands in hot weather and contracts in cold weather. Gaps allow space for expansion and prevent the floor from cracking.
Question 9
Why is one end of a steel girder in a bridge kept on rollers instead of fixing it in a pillar?
Answer:
One end of a steel girder in a bridge is kept on rollers to allow for thermal expansion and contraction. When temperature rises, the steel girder expands, and when temperature falls, it contracts. If both ends were fixed, the expansion would produce large stresses that could bend or crack the bridge. Rollers allow the girder to move slightly, preventing damage.
Question 10
An empty glass bottle is fitted with a narrow tube at its mouth. The open end of the tube is kept in a beaker containing water. When the bottle is heated, bubbles of air are seen escaping into water. Explain the reason.
Answer:
When the bottle is heated, the air inside the bottle gets heated and expands. Due to expansion, the pressure of air inside the bottle increases, forcing some of the air to escape through the narrow tube into the water, which appears as bubbles.
Question 11
An iron washer is heated. State the effect on its:
(a) mass
(b) internal diameter
(c) external diameter and
(d) density.
Answer:
(a) Mass remains unchanged, because no material is lost or gained.
(b) The internal diameter increases. On heating, the washer expands in all directions, including the hole.
(c) The external diameter increases due to thermal expansion.
(d) Density decreases, because the volume increases while the mass remains constant.
Download Heat Transfer Solutions PDF
You can also download the PDF version for offline study and revision anytime. It is helpful during exam time when internet is not available.
Conclusion
If you want to score high in Physics, practice regularly with ICSE Class 8 Physics Chapter 6 Heat Transfer Selina Solutions. These solutions make learning easy and help you understand every concept clearly. Keep revising and solve all textbook questions for best results.
ICSE Class 8 Physics Selina Solutions
The Selina Concise Physics Class 8 book includes the following chapters:
Official Website:
Students can visit the official CISCE website for more details and updates.



