Introduction
If you are searching for ICSE Class 8 Physics Matter Selina Solutions, you are in the right place. This chapter is one of the most important topics in Class 8 Physics because it explains the concept of matter, its states, properties, and changes. Students often look for accurate answers to textbook exercises for homework, revision, and exam preparation.
In this article, we provide easy and clear Selina Solutions Class 8 Physics Matter to help students understand every concept step by step.
Study Support by Rohit Academy
Rohit Academy offers expert-curated ICSE Class 8 Physics Study Materials including ICSE Matter Chapter Notes, diagrams, and key formulas for better understanding.
You can also visit:
| ☛ ICSE Class 8 Physics Chapter 1: Matter Notes |
| ☛ ICSE Class 8 Physics |
| ☛ ICSE Class 8 Chemistry |
| ☛ ICSE Class 8 Mathematics |
| ☛ ICSE Class 8 Biology |
(A) Multiple Choice Type
Question 1
Choose the correct answer from the multiple choices given below.
Question 1(a)
The inter-molecular force is maximum in:
(i) solids (ii) gases
(iii) liquids (iv) none of the above
Answer:
(i) solids
Explanation:
In solids, particles are tightly packed and strongly attracted to each other, so intermolecular forces are strongest.
Question 1(b)
The inter-molecular space is maximum in:
(i) liquids (ii) solids
(iii) gases (iv) none of the above
Answer:
(iii) gases
Explanation:
Gas particles are far apart, so the space between molecules is maximum.
Question 1(c)
The molecules can move freely anywhere in
(i) gases (ii) liquids
(iii) solids (iv) none of the above
Answer:
(i) gases
Explanation:
In gases, particles have very weak forces of attraction and can move freely in all directions.
Question 1(d)
The molecules move only within the boundary in :
(i) liquids (ii) gases
(iii) solids (iv) none of the above
Answer:
(i) liquids
Explanation:
In liquids, particles can move but are still confined within the container’s boundaries.
Question 1(e)
The temperature at which a liquid gets converted into its vapour state is called its:
(i) melting point (ii) boiling point
(iii) dew point (iv) freezing point
Answer:
(ii) boiling point
Explanation:
Boiling point is the temperature at which a liquid changes into vapour throughout the liquid.
Question 1(f)
Rapid conversion of water into steam is an example of:
(i) evaporation (ii) freezing
(iii) melting (iv) vaporization
Answer:
(iv)vaporization
Explanation:
Vaporization (boiling) is a rapid process where liquid turns into gas.
Question 1(g)
Evaporation takes place from the:
(i) from the surface of liquid
(ii) throughout the liquid
(iii) from the mid-portion of the liquid
(iv) from the bottom of liquid
Answer:
(i) from the surface of liquid
Explanation:
Evaporation is a slow process that occurs only at the surface.
Question 1(h)
Boiling takes place from:
(i) from the surface of the liquid
(ii) throughout the liquid
(iii) from mid-portion of liquid
(iv) from none of the above
Answer:
(ii) throughout the liquid
Explanation:
During boiling, bubbles form throughout the liquid, not just at the surface.
Question 1(i)
Identify the correct statement.
(1) The molecules of water move faster than ice.
(2) The molecules are more closely packed in ice than water.
(3) The molecules of water vibrate to and fro only but those of ice are free to move within the boundary of the vessel in which it is kept.
(4) The molecules have more intermolecular space in ice than in water.
(i) (1), (2) and (3)
(ii) (2), (3) and (4)
(iii) (1) and (2)
(iv) all are correct
Answer:
(iii) (1) and (2)
Assertion Reason Type
Question 2
The following questions are Assertion-Reason based questions. Choose the answer based on the codes given below.
Question 2(a)
Assertion (A) : We often pour tea in a saucer to cool it faster.
Reason (R) : Evaporation takes place at all temperatures.
(i) Both A and R are true and R is the correct explanation of A
(ii) Both A and R are true and R is not the correct explanation of A
(iii) Assertion is true but reason is false
(iv) Assertion is false but reason is true
Answer:
(ii) Both A and R are true and R is not the correct explanation of A
Explanation:
Both are true, but cooling is mainly due to increased surface area, not just evaporation at all temperatures.
Question 2(b)
Assertion (A) : The molecules in a liquid can move within the boundary of the vessel.
Reason (R) : A liquid has a definite volume and a definite shape.
(i) Both A and R are true and R is the correct explanation of A
(ii) Both A and R are true and R is not the correct explanation of A
(iii) Assertion is true but reason is false
(iv) Assertion is false but reason is true
Answer:
(iii) Assertion is true but reason is false
Explanation:
Assertion is true, but liquids do not have definite shape, so reason is false.
Fill in the blanks
Question 3
Fill in the blanks:
(a) Evaporation takes place at …………… temperatures.
(b) …………… process is just the reverse of melting.
(c) …………… is a process that involves direct conversion of a solid into its vapour on heating.
(d) The temperature at which a solid converts into a liquid is called its …………… .
(e) The smallest unit of matter that exists freely in nature is called …………… .
(f) Molecules of a substance are always in a state of …………… and so they possess …………… .
(g) Inter-molecular space is maximum in …………… less in …………… and the least in …………… .
(h) Inter-molecular force of attraction is maximum in …………… , less in …………… and the least in …………… .
Answer:
(a) Evaporation takes place at all temperatures.
(b) Freezing process is just the reverse of melting.
(c) Sublimation is a process that involves direct conversion of a solid into its vapour on heating.
(d) The temperature at which a solid converts into a liquid is called its melting point.
(e) The smallest unit of matter that exists freely in nature is called molecule.
(f) Molecules of a substance are always in a state of motion and so they possess kinetic energy.
(g) Inter-molecular space is maximum in gases less in liquids and the least in solids.
(h) Inter-molecular force of attraction is maximum in solids, less in liquids and the least in gases.
True or False
Question 4
Write true of false for each statement :
(a) The temperature of a substance remains unaffected during its change of state.
(b) Ice melts at 100°C.
(c) Water at 100°C has more heat than steam at 100°C.
(d) Evaporation of a liquid causes cooling.
(e) Water evaporates only at 100°C.
(f) Boiling takes place at all temperatures.
(g) Evaporation takes place over the entire mass of the liquid.
(h) The process of a gas converting directly into solid is called vaporization.
(i) At high altitudes, water boils above 100°C.
(j) The melting point of ice is 0°C.
Answer:
(a) True
(b) False
Correct Statement : Ice melts at 0°C.
(c) False
Correct Statement : Steam at 100°C has more heat than water at 100°C.
(d) True
(e) False
Correct Statement : Water evaporates at all temperatures.
(f) False
Correct Statement : Boiling takes place at a fixed temperature which is 100°C.
(g) False
Correct Statement : Evaporation takes place only at the surface of the liquid.
(h) False
Correct Statement : The process of a gas converting directly into solid is called deposition or solidification.
(i) False
Correct Statement : At high altitudes, water boils below 0°C.
(j) True
Question 5
Match the following:
| Column A | Column B |
| (a) Molecules | (i) water boils |
| (b) 100°C | (ii) evaporation |
| (c) 0°C | (iii) changes from solid to gas |
| (d) At all temperatures | (iv) matter |
| (e) Camphor | (v) water freezes |
Answer:
| Column A | Column B |
| (a) Molecules | (iv) matter |
| (b) 100°C | (i) water boils |
| (c) 0°C | (v) water freezes |
| (d) At all temperatures | (ii) evaporation |
| (e) Camphor | (iii) changes from solid to gas |
(B) Short Answer Type Questions
Question 1
Define the term matter. What is it composed of?
Answer:
Matter is anything that has mass and occupies space.
It is composed of tiny particles called molecules (or atoms).
Question 2
State three properties of molecules of matter.
Answer:
Three properties of molecules of matter:
1. They have spaces between them.
2. They are in constant motion.
3. They attract each other.
Question 3
What do you mean by inter-molecular spaces? How do they vary in different states of matter?
Answer:
Inter-molecular spaces are the spaces between molecules.
● Solids → very small
● Liquids → moderate
● Gases → very large
Question 4
What is meant by inter-molecular forces of attraction? How do they vary in solids, liquids and gases?
Answer:
Inter-molecular forces of attraction are forces that hold molecules together.
● Solids → strongest
● Liquids → weaker
● Gases → weakest
Question 5
Which of the following are correct?
(a) Solids have definite shape and definite volume.
(b) Liquids have definite volume but no definite shape.
(c) Gases have definite volume but no definite shape.
(d) Liquids have both definite shape and definite volume.
Answer:
The correct options are:
(a) Solids have definite shape and definite volume.
Explanation: In solids, the intermolecular space is very small and the force of attraction between molecules is very strong. As a result, the molecules remain fixed in their positions and can only vibrate. Therefore, solids have a definite shape as well as a definite volume.
(c) Liquids have definite volume but no definite shape.
Explanation: In liquids, the molecules are less closely packed and the intermolecular force of attraction is weaker than in solids. The molecules can move freely but remain within the container due to some attraction. Hence, liquids have a definite volume but take the shape of the container.
Question 6
A liquid can change into vapour state
(a) at a fixed temperature and
(b) at all temperatures
Name the processes involved in the two cases.
Answer:
(a) At fixed temperature → Boiling (Vaporization)
(b) At all temperatures → Evaporation
Question 7
State
(a) the melting point of ice and
(b) the boiling point of water
Answer:
(a) The melting point of ice is 0 °C.
(b) The boiling point of water is 100 °C.
Question 8
What is evaporation?
Answer:
Evaporation is the slow conversion of a liquid into vapour from its surface at any temperature.
Question 9
State three factors which affect the rate of evaporation of a liquid.
Answer:
Factors affecting evaporation:
1. Temperature
2. Surface area
3. Wind speed (air flow)
Question 10
Why are volatile liquids such as alcohol and spirit stored in tightly closed bottles?
Answer:
Volatile liquids are stored in tightly closed bottles to prevent evaporation and loss of liquid.
Question 11
Why is cooling produced on evaporation of a liquid?
Answer:
During evaporation, a liquid changes into gas by absorbing heat from its surroundings. This absorption of heat lowers the temperature of the surroundings, producing a cooling effect.
Question 12
Give two applications of evaporation.
Answer:
Applications of evaporation:
1. Drying of clothes
2. Making salt from seawater
Question 13
What do you mean by sublimation? Explain with an example.
Answer:
Sublimation is the direct change of solid into vapour without becoming liquid.
Example: Naphthalene balls
Question 14
Why does the size of naphthalene balls decrease when left open?
Answer:
Naphthalene balls decrease in size because they sublime (change directly into vapour) when left open.
(C) Long Answer Type Questions
Question 1
Discuss the three states of matter: solid, liquid and gas on the basis of molecular model.
Answer:
1. Solid
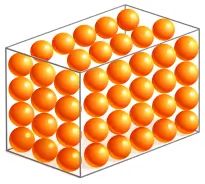
In solids, molecules are very closely packed and arranged in a fixed, orderly pattern.
❖ Molecular Characteristics:
- Intermolecular force: Very strong
- Space between molecules: Very small
- Movement: Only vibrate about fixed positions
- Arrangement: Regular and fixed
- Properties: Definiteshape and definite volume.
2. Liquid
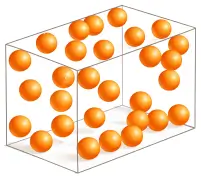
In liquids, molecules are close together but not fixed. They can move and slide past each other.
❖ Molecular Characteristics:
- Intermolecular force: Moderate
- Space between molecules: More than solids
- Movement: Free to move (flow)
- Arrangement: Irregular
- Properties: No definite shape (takes container shape), but definite volume.
3. Gas
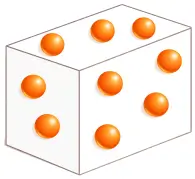
In gases, molecules are far apart and move freely and rapidly in all directions.
❖ Molecular Characteristics:
- Molecular Arrangement: Molecules are very far apart.
- Structure: No ordered structure.
- Movement: Molecules move fast, randomly, and in all directions.
- Forces: Intermolecular forces are negligible.
- Space between molecules: Maximum
- Properties: No definite shape and no definite volume
Question 2
What do you mean by ‘the change of state’? Write the flowchart showing the complete cycle of change of state.
Answer:
The conversion of a substance from one physical state to another, either by absorbing or releasing heat while the temperature remains constant, is known as change of state.
The complete cycle of change of state can be represented by the following flowchart:
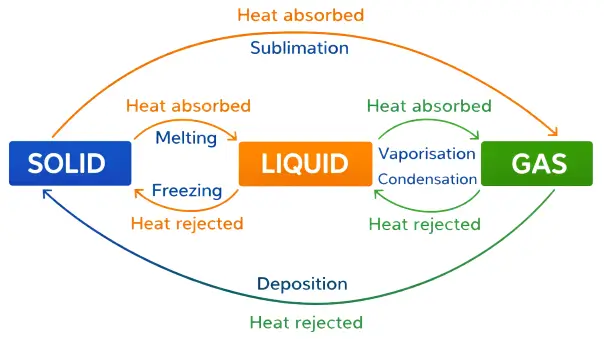
Question 3
Differentiate between melting point and boiling point, giving atleast one example of each.
Answer:
| Melting Point | Boiling Point |
| Melting point is the temperature at which a solid changes into a liquid on heating. | Boiling point is the temperature at which a liquid changes into gas throughout the liquid. |
| Ice melts into water at 0°C (273 K) | Water boils into steam at 100°C (373 K) |
Question 4
Describe the process of condensation and sublimation with examples.
Answer:
Condensation:
- Condensation is the process in which a gas changes into a liquid on cooling.
- Examples: Formation of water droplets on the outside of a cold glass
Sublimation:
- Sublimation is the process in which a solid directly changes into gas without becoming liquid.
- Examples: Dry ice (solid CO2) changing directly into gas
Question 5
Explain the terms melting and melting point.
Answer:
Melting:
Melting is the process by which a solid changes into a liquid on heating.
Melting Point:
Melting point is the fixed temperature at which a solid changes into a liquid at atmospheric pressure.
Example: Ice melts into water at 0°C (273 K).
Question 6
Describe an experiment to demonstrate that a substance absorbs heat during melting without change in its temperature.
Answer:
The following experiment shows that a substance absorbs heat during melting without any change in temperature.
Experiment:
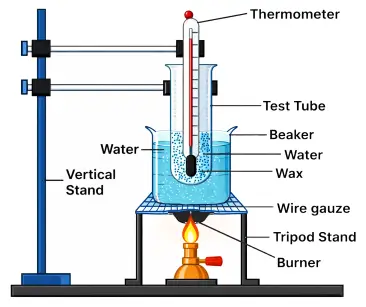
Take a test tube containing some wax. Fix the test tube vertically on a stand and place it inside a beaker filled with water. Keep the beaker on a wire gauze placed over a tripod stand.
Now clamp a thermometer on the same stand and insert it into the test tube so that its bulb remains inside the wax. Heat the beaker gently using a burner. Record the temperature of the wax at regular intervals (every minute).
Observations:
The temperature of wax rises steadily at first. When it reaches 55°C, the temperature remains constant for some time, even though heating is continued. After all the wax melts, the temperature again starts rising.
Conclusion:
Wax melts at 55°C. During the process of melting, heat is continuously supplied but the temperature does not increase. This shows that the heat is absorbed as latent heat of fusion, proving that a substance absorbs heat during melting without any change in temperature.
Question 7
Explain the terms vaporization and boiling point.
Answer:
Vaporization:
Vaporization is the process by which a liquid changes into vapour (gas) on heating.
Boiling Point:
Boiling point is the fixed temperature at which a liquid changes into vapour throughout the liquid at atmospheric pressure.
Example:
Water boils and changes into steam at 100°C (373 K).
Question 8
Describe an experiment to demonstrate that water absorbs heat during boiling at a constant temperature.
Answer:
Aim: To show that water absorbs heat during boiling without any rise in temperature.
Apparatus: Beaker, water, thermometer, tripod stand, wire gauze, burner.
Procedure:
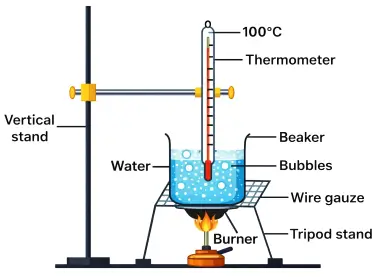
- Take some water in a beaker.
- Clamp a thermometer so that its bulb is immersed in the water.
- Place the beaker on a wire gauze over a tripod stand.
- Heat the water using a burner.
- Record the temperature at regular intervals (every minute).
Observation:
- The temperature of water rises gradually up to 100°C.
- At 100°C, water starts boiling.
- Even on continuous heating, the temperature remains constant at 100°C until all the water changes into steam.
Conclusion:
Water absorbs heat during boiling without any change in temperature. This heat is used to convert liquid into vapour and is called latent heat of vaporization.
Result:
A liquid absorbs heat during boiling at a constant temperature.
Question 9
Wet clothes dry more quickly on a warm dry day than on a cold humid day. Explain.
Answer:
Evaporation depends on temperature and the amount of moisture (humidity) present in the air. The rate of evaporation is higher in dry air than in humid air and also increases with a rise in temperature. Therefore, wet clothes dry faster on a warm dry day than on a cold humid day.
Question 10
Explain with an example that when a liquid evaporates, it takes heat from its surroundings.
Answer:
When a liquid evaporates, it absorbs heat from its surroundings to change into vapour. This causes a cooling effect.
Example:
If a few drops of spirit or alcohol are placed on the palm, it feels cool. This is because the liquid evaporates by taking heat from the skin, thereby lowering its temperature.
Question 11
Explain why in hot summer days water remains cool in earthen pots?
Answer:
Earthen pots have tiny pores through which water seeps out and evaporates. During evaporation, heat is absorbed from the water inside, making it cool. Hence, water remains cool in earthen pots.
Question 12
Describe an experiment to demonstrate the process of sublimation.
Answer:
Aim:
To demonstrate the process of sublimation.
Apparatus:
China dish, ammonium chloride (or iodine), inverted funnel, cotton plug, tripod stand, burner.
Procedure:
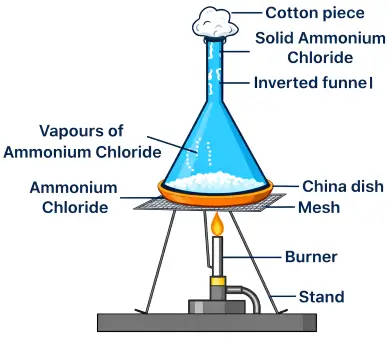
Take a small amount of ammonium chloride in a china dish.
Cover it with an inverted funnel and plug the stem of the funnel with cotton.
Place the china dish on a tripod stand and heat it gently using a burner.
Observation:
- The solid ammonium chloride changes directly into vapours on heating.
- These vapours rise and cool on the inner surface of the funnel, forming solid crystals again.
Conclusion:
This shows that ammonium chloride changes directly from solid to vapour and back to solid without passing through the liquid state, which is called sublimation.
(D) Think and Answer
Question 1
Some ice is taken in a beaker and its temperature is recorded after each one minute. The observations are listed below:
| Time (in minute) | Temperature (in °C) |
| 0 | 0 |
| 1 | 0 |
| 2 | 0 |
| 3 | 0 |
| 4 | 0 |
| 5 | 0 |
| 6 | 3.8 |
| 7 | 7.6 |
| 8 | 11.4 |
From the above observations what conclusion do you draw about the melting point of the ice?
Answer:
From the above observations, it is concluded that the melting point of ice is 0°C.
At this temperature, ice begins to melt by absorbing heat without any increase in temperature. Once all the ice has melted into water (after about 5 minutes), the temperature of the water starts rising further.
Question 2
Water in a dish evaporates faster than in a bottle. Give reason.
Answer:
Water in a dish has a larger exposed surface area, so evaporation is faster. In a bottle, the surface area is smaller, so evaporation is slower.
Question 3
A certain quantity of water is heated from 20°C to 100°C. Its temperature is recorded after each 1 minute. The observations are:
| Time (in minute) | Temperature (in °C) |
| 0 | 20 |
| 1 | 30 |
| 2 | 40 |
| 3 | 50 |
| 4 | 60 |
| 5 | 70 |
| 6 | 80 |
| 7 | 90 |
| 8 | 100 |
| 9 | 100 |
| 10 | 100 |
| 11 | 100 |
| 12 | 100 |
What conclusion do you draw from the above table about the boiling point of water? Explain.
Answer:
From the above observations, it is concluded that the boiling point of water is 100°C.
At this temperature, water starts boiling and changes into steam by absorbing the heat supplied, without any further increase in temperature.
Question 4
A patient suffering from high fever is advised to put wet cloth strips on his forehead. Why?
Answer:
Wet cloth strips contain water which evaporates from the patient’s forehead. During evaporation, water absorbs heat from the body, thereby lowering the body temperature.
Hence, wet cloth strips help in reducing high fever by cooling the body.
Question 5
When we put some acetone or petrol on our palm, it feels cold. Give reason.
Answer:
Acetone or petrol evaporates very quickly when placed on the palm. During evaporation, it absorbs heat from the skin (latent heat of vaporization), which lowers the temperature of the palm and produces a cooling sensation.
Question 6
On a humid day, you sweat more and feel less cool in comparison to a breezy day where sweating makes you feel much cooler. Give reason.
Answer:
On a humid day, air contains more moisture, so sweat evaporates slowly and less cooling occurs. On a breezy day, evaporation is faster due to moving air, so more heat is removed and we feel cooler.
ICSE Class 8 Physics Matter Selina Solutions PDF Download
Download ICSE Class 8 Physics Matter Selina Solutions PDF with chapter-wise solved answers, explanations, and free study resources for easy exam preparation.
Why Study ICSE Class 8 Physics Matter Selina Solutions?
Using ICSE Class 8 Physics Matter Selina Solutions helps students:
- Understand textbook questions easily
- Learn correct exam answers
- Improve concept clarity
- Save revision time
- Score better marks in exams
Exam Tips for Matter Chapter
- Learn all definitions carefully.
- Understand differences between solid, liquid, and gas.
- Practice diagrams and examples.
- Revise change of state processes.
- Solve textbook exercises regularly.
Conclusion
This chapter is the foundation of physics and chemistry. By using these ICSE Class 8 Physics Matter Selina Solutions, students can prepare confidently for school tests and annual exams. Keep practicing chapter questions regularly for the best results.
If you need complete exercise-wise answers, MCQs, numericals, or notes, bookmark this page for future learning.
ICSE Class 8 Physics Selina Solutions
The Selina Concise Physics Class 8 book includes the following chapters:
Official Website:
Students can visit the official CISCE website for more details and updates.


