ICSE Class 8 Chemistry Chapter 1: Matter Notes
Are you searching for ICSE Class 8 Chemistry Matter Notes PDF Download? Here you will get complete and easy-to-understand ICSE Class 8 Chemistry Matter Notes prepared according to the latest syllabus. These notes help students revise important concepts quickly before exams.
Matter is one of the most important chapters in ICSE Class 8 Chemistry. It explains the meaning of matter, states of matter, interconversion of states, physical and chemical changes, evaporation, melting, boiling, condensation, and freezing.
Study Support by Rohit Academy
Rohit Academy offers expert-curated ICSE Class 8 Chemistry Study Materials including ICSE Measurements and Experimentation Chapter Notes, diagrams, and key formulas for better understanding.
You can also visit :
| ☛ ICSE Class 8 Chemistry Chapter 1: Matter Selina Solutions |
| ☛ ICSE Class 8 Physics |
| ☛ ICSE Class 8 Chemistry |
| ☛ ICSE Class 8 Mathematics |
| ☛ ICSE Class 8 Biology |
Class 8 Chemistry Matter Notes ICSE
- Matter: Matter is anything that has mass and occupies space.
Examples: Iron, Wood, Book, Water, Oil, air, oxygen etc.
***Note
Air is also matter because it occupies space.
Composition of Matter
- Greek philosophers believed matter is made of four elements: fire, water, air, and earth.
- Ancient Indian philosophers believed matter is made of five elements (tatvas): Akash (space), Vayu (air), Tejas (fire), Jal (water), and Prithvi (earth).
- Maharshi Kanada suggested that matter is made of tiny particles called anu, further made of paramanu.
- Democritus called the smallest particle atom.
- John Dalton later proposed that matter is made of very small particles called atoms.
Atom:
- An atom is the smallest particle of matter that exhibits all the properties of that matter.
- Atoms usually do not exist independently. They combine with each other to form molecules.
Molecule:
A molecule is a group of atoms joined together and can exist independently.
Kinetic Theory of Matter
The kinetic theory of matter explains:
- The existence of the three states of matter
- Their properties
- The interconversion of matter from one state to another
Main Postulates
- Matter is made up of very tiny particles called atoms or molecules.
- The particles of matter are in constant motion. They keep moving continuously in all directions.
- There are spaces between the particles of matter, known as intermolecular spaces.
- The particles of matter attract each other. This attraction is called intermolecular force of attraction.
- The particles possess kinetic energy, and this energy increases when the temperature increases.
Brownian Motion
Brownian motion is the random zig-zag movement of tiny particles in a liquid or gas.
Diffusion
Diffusion is the intermixing of two or more substances due to the motion of their particles to form a uniform mixture.
Examples:
- Smell of perfume spreading in a room
- Ink mixing in water
Diffusion is:
- Fastest in gases
- Slower in liquids
- Very slow in solids
Intermolecular Space
- The spaces between particles of matter are called intermolecular spaces.
- Example: When salt dissolves in water, the water level does not increase because salt particles occupy spaces between water molecules.
Intermolecular Force of Attraction
Particles of matter attract each other. This force is called intermolecular force of attraction.
The strength of this force varies in different substances.
Kinetic Energy
The energy possessed by a body due to its motion is called kinetic energy.
Particles of matter have kinetic energy because they are always moving.
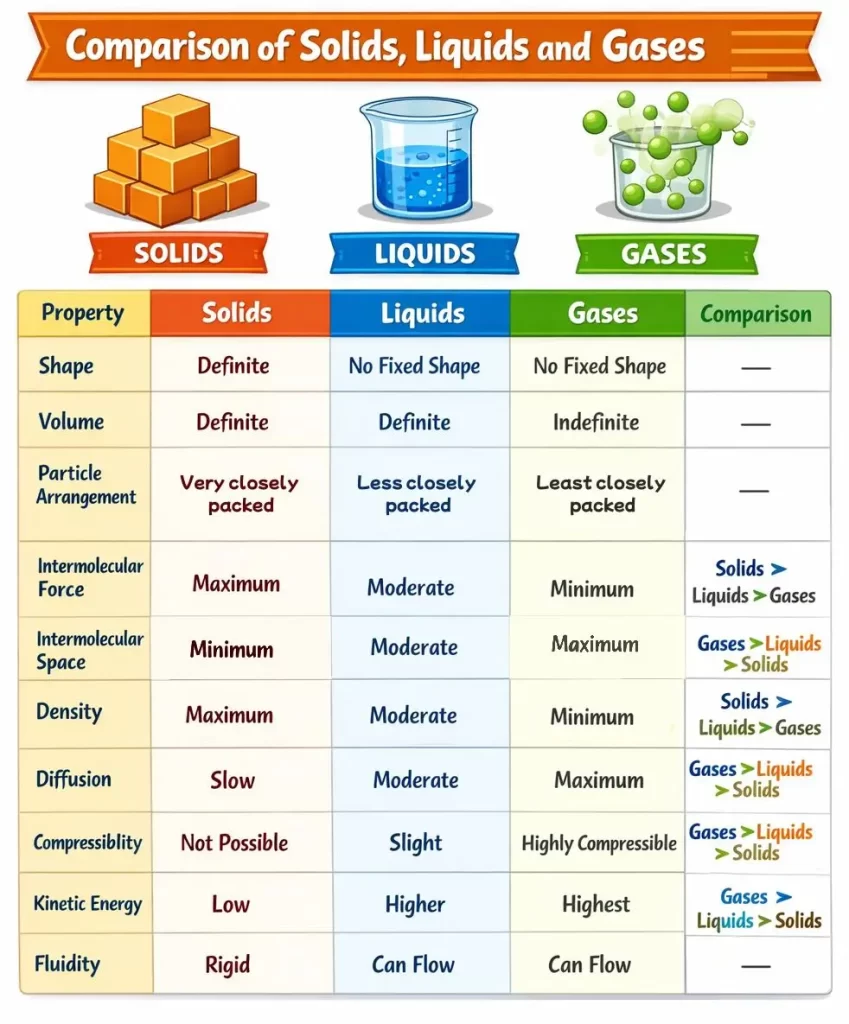
States of Matter
Matter exists in three main states:
- Solid
- Liquid
- Gas
Solids
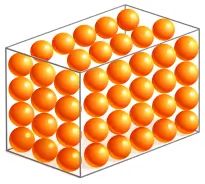
- Have definite shape and definite volume
- Particles are very closely packed
- Strong intermolecular forces
- Very little intermolecular space
- Rigid and difficult to compress
- Lowest kinetic energy
- Examples: Wood, iron, ice, stone
Liquids
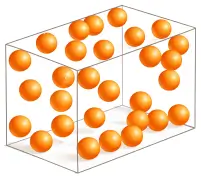
- Have definite volume but no fixed shape
- Take the shape of the container
- Particles are loosely packed
- Moderate intermolecular forces
- Can flow
- Higher kinetic energy than solids
- Examples: Water, milk, alcohol, oil
Gases
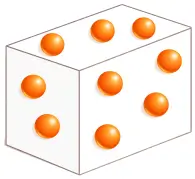
- Have no definite shape and no definite volume
- Fill the entire container
- Particles are far apart
- Very weak intermolecular forces
- Highly compressible
- Highest kinetic energy
- Examples: Air, oxygen, hydrogen, water vapour
Fluids
- Substances that can flow are called fluids.
- Liquids and gases are fluids.
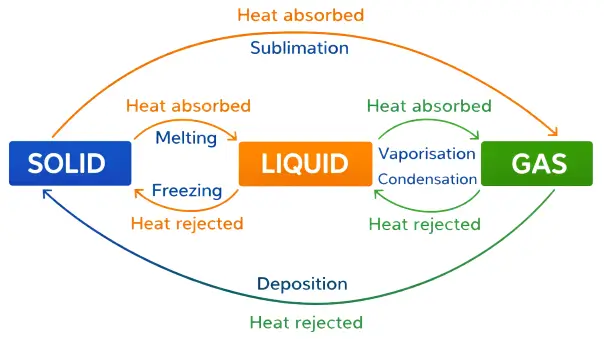
Interconversion of States of Matter
The change of matter from one state to another and back again without changing its chemical composition is called interconversion of states of matter.
Causes of Change of State:
- Change in temperature
- Change in Pressure
Change of State by Changing Temperature
- When a substance in solid state is heated, it changes into a liquid.
- On further heating, the liquid changes into a gas.
- Reverse changes occur when the substance is cooled.
Examples of Change of State
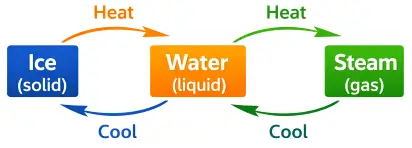
1. Water: Ice melts into water on heating and water changes into steam on boiling.
2. Candle wax: Solid wax melts into liquid wax on heating and solidifies again on cooling.
How and Why Do These Changes Take Place?
- On heating, the kinetic energy of its particles increases. The particles move faster and farther apart. The intermolecular forces of attraction become weaker.
Example: Solid → Liquid → Gas - On cooling, the kinetic energy of its particles decreases. The particles move more slowly and come closer together. The intermolecular forces of attraction become stronger.
Example: Gas → Liquid → Solid
Sublimation
- The process in which a solid directly changes into gaseous state without passing through the liquid state is called sublimation.
- Examples: Camphor, Iodine, Naphthalene, Ammonium chloride, Dry ice (solid CO2) etc.
Deposition
The process in which a gas directly changes into solid state without passing through the liquid state is called deposition.
Why Do Certain Solids Sublime?
Certain solids have weaker intermolecular forces of attraction compared to other solids.
When heated, their particles quickly gain kinetic energy and change directly into vapour.
Change of State of Matter by Changing Pressure
- Pressure is also an important factor that causes change in the state of matter.
- A gas can be changed into a liquid by cooling and increasing pressure.
- Examples
◉ Liquid oxygen and liquid hydrogen are obtained by increasing pressure and lowering temperature.
◉ LPG cylinders contain cooking gas in liquid form under high pressure.
Terms Related to Change of State
- Melting or Fusion: The process by which a solid changes into liquid state on heating is called melting or fusion.
- Melting Point: The fixed temperature at which a solid changes into liquid at atmospheric pressure is called its melting point.
Example: The melting point of ice is 0°C.
- Freezing (Solidification): The process by which a liquid changes into solid state is called freezing or solidification.
- Freezing Point: The temperature at which a liquid starts changing into solid on cooling is called its freezing point.
Example: Pure water freezes at 0°C under normal conditions.
- Evaporation (Vaporisation): The process by which a liquid changes into vapour state is called evaporation or vaporisation.
◉ Evaporation can occur at room temperature.
◉ The rate of evaporation increases with heating.
- Boiling Point: The fixed temperature at which a liquid changes into vapour state on heating is called its boiling point.
Example: The boiling point of pure water is 100°C.
- Condensation: The process by which a gas changes into liquid state is called condensation or liquefaction.
- Condensation Point: The temperature at which a gas changes into liquid on cooling.
Example: The condensation point of steam is 100°C.
- Liquefaction: The process of converting a gas into liquid state by applying pressure and lowering temperature is called liquefaction.
Law of Conservation of Mass
Matter can neither be created nor destroyed in a chemical reaction.
This means:
Total mass of reactants = Total mass of products
This law was proposed by Antoine Lavoisier.
- Example 1
When barium chloride solution reacts with sodium sulphate solution, a white precipitate of barium sulphate is formed.
Chemical reaction:
BaCl₂ (aq) + Na₂SO₄ (aq) → BaSO₄ (s) + 2NaCl (aq) When the reaction is carried out in a closed flask, the mass of the apparatus before and after the reaction remains the same. This verifies the Law of Conservation of Mass.
- Example 2
When baking soda (sodium bicarbonate) reacts with vinegar (acetic acid), carbon dioxide gas is produced, causing fizzing.
The reaction is:
NaHCO₃ + CH₃COOH → CO₂ + CH₃COONa + H₂O
When the experiment is done in a closed container, the total mass of reactants equals the total mass of products, verifying the Law of Conservation of Mass.
- Example 3: Burning of Wood
When wood burns, ash is formed.
The mass of ash appears less than the mass of wood because gaseous products (like smoke and water vapor) are released into the air and not weighed.
- Example 4: Burning of Magnesium
When magnesium ribbon burns in air, magnesium oxide is formed.
Chemical reaction:
2Mg + O₂ → 2MgO
The mass of magnesium oxide appears more than magnesium because magnesium combines with oxygen from the air to form magnesium oxide.
If oxygen is considered, the total mass of reactants and products is equal.
ICSE Class 8 Chemistry Matter Notes PDF Download
Students searching for ICSE Class 8 Chemistry Matter Notes PDF Download can save this page for revision or convert it into PDF for offline study. These notes are designed in simple language for better understanding.
Importance of Matter Notes for Exams
These ICSE Class 8 Chemistry Matter Notes PDF Download are useful for:
- Quick revision before exams
- Understanding basic concepts
- Solving textbook questions
- Preparing school tests
Conclusion
These ICSE Class 8 Chemistry Matter Notes are designed in simple language for students. Learn definitions, examples, and important differences to score better marks in exams. Save these notes for regular revision and better preparation.
ICSE Class 8 Chemistry Notes
| ☛ ICSE Class 8 Chemistry Chapter 1 – Matter Notes |
| ☛ ICSE Class 8 Chemistry Chapter 2 – Physical and Chemical Changes Notes |
| ☛ ICSE Class 8 Chemistry Chapter 3 – Elements, Compounds and Mixtures Notes |
| ☛ ICSE Class 8 Chemistry Chapter 4 – Atomic Structure Notes |
| ☛ ICSE Class 8 Chemistry Chapter 5 – Language of Chemistry Notes |
| ☛ ICSE Class 8 Chemistry Chapter 6 – Chemical Reactions Notes |
| ☛ ICSE Class 8 Chemistry Chapter 7 – Hydrogen Notes |
| ☛ ICSE Class 8 Chemistry Chapter 8 – Water Notes |
| ☛ ICSE Class 8 Chemistry Chapter 9 – Carbon and its Compounds Notes |
Official Website:
Students can visit the official CISCE website for more details and updates.


